


WHY USE POLAR BODIES FOR ANEUPLOIDY AND
CYSTIC FIBROSIS DIAGNOSIS FOR PRE-IMPLANTATION
DIAGNOSIS IN GERMANY?
Schwinger E1,*, Tomi D1, Voigt R1, Eckhold J1, Hinrichs F1, Diedrich K2
*Corresponding Author: Professor Dr. (med.) E. Schwinger, Institute of Human Genetics, Medical University of Lübeck, Ratzeburger Allee 160, 23538 Lübeck, Germany; Tel.: +49(0)451-500-2621; Fax: +49(0)451-500-4187; E-mail: schwing@uni-luebeck.de
page: 15
|
|
INTRODUCTION
After assisted reproduction, pre-implantation genetic diagnosis (PGD) enables the detection of certain genetic disorders in embryos in vitro before they are transferred into the uterus. This technique allows for the analysis of a known genetic disorder in offspring of couples with a predisposition for these disorders. Medical and psychological complications associated with a possible termination of the pregnancy after prenatal diagnosis can be reduced by this technique.
In Germany, PGD is not allowed because of the Embryo Protection Law (Embryonenschutzgesetz). According to this law, it is only allowed to fertilize oocytes with the aim of establishing a pregnancy. This law defines an embryo in that moment the two pronuclei are fused. After fusion of the pronuclei it is not allowed to discard the embryo. The use of the embryo for other purposes is forbidden. Therefore, scientific investigation or diagnosis in blastomeres, as well as the donation of oocytes, is illegal in Germany.
Pre-implantation genetic diagnosis, usually performed by testing blastomeres, enables couples to minimize the individual risk for known molecular genetic or chromosomal disorders. In Germany, pregnancy termination after prenatal diagnosis of chromosomal aberrations or genetic diseases is tolerated, while PGD is prohibited. The genetic disease of the fetus itself is not sufficient reason to justify an abortion. The present and the future situation of the mother with possible clinical or psychological affections has to be taken into consideration. There are couples who decide to continue pregnancies after prenatal diagnosis of trisomy 21 or cystic fibrosis (CF).
The investigation of polar bodies, according to the definition of an embryo by German law, is the only possible way to perform PGD in Germany. The investigation of polar bodies has to be finished prior to the fusion of the pronuclei. Pre-implantation genetic diagnosis using polar bodies is called pre-fertilization diagnosis (PFD). Pre-fertilization diagnosis can be performed for molecular and cytogenetic disorders [1,2]. We established PFD for CF using multiplex polymerase chain reaction (PCR) and for aneuploidy diagnosis using fluorescence in situ hybridization (FISH) in the Department of Human Genetics at Lübeck, Germany (see also Pre-implantation genetic diagnosis using polar bodies in this issue). Multiplex PCR for molecular genetic disorders is necessary in polar bodies to avoid misdiagnosis due to a crossover in the first meiotic division.
The applied techniques for molecular as well as for molecular cytogentic diagnosis are time consuming. Due to the restrictions of German law there is only a short time span to perform PFD (Fig. 1). The available time for diagnosis using the first polar body is 24 hours, for investigation of the second polar body only 10-16 hours. After recognizing the pronuclei there are only 4 to 8 hours left until cryopreservation has to be accomplished. For oocytes with polar body biopsie the chance of embryo development after cryopreservation is reduced. Because of this, PFD and oocyte transfer in the same cycle is desirable.
In Germany, scientists and people affected by genetic diseases in their family are trying to influence political opinion to change the legal situation. There are also adverse opinions from the clerical side as well as from organizations like self-supporting groups. In Germany, the self-supporting groups for different diseases are well organized and play an active role in influencing public opinion for prenatal and PGD as well as for pregnancy termination. The increased life-span of children with trisomy 21 and with CF so that the attitude towards abortion is very critical in Germany. Self-supporting groups worry about discrimination against ill or handicapped persons due to pregnancy termination in cases of genetic diseases. There are fears that PGD underlines selection and increases discrimination against handicapped people.
There are some disadvantages to using polar bodies for PGD of molecular genetic disorders. Since only the genetic traits of the mother can be tested, polar body diagnosis is not suitable for detecting autosomal dominant disorders inherited by and from the father. In cases of recessive disorders, all oocytes carrying the gene defect have to be discarded, even though only 50% develop the autosomal recessive disease after fertilization. Some X-linked diseases may have a mild phenotype in women, but the gender of the embryo is determined by the X or Y chromosome in the sperm. Performing PGD all oocytes carrying a mutation have to be discarded. A further difficulty in analyzing polar bodies is caused by crossover prior the first meiotic division, leading to heterozygous first polar bodies. Due to the high percentage of heterozygosity in first polar bodies [1], the second polar body should be tested to predict whether the oocyte is free of the tested mutation.
For analyzing cytogenetic disorders such as translocations or numerical chromosomal anomalies polar body diagnosis seems to be an alternative to blastomere investigation. Polar body diagnosis for translocations is, of course, only practicable if the translocation is transmitted by the mother. Aneuploidy diagnosis using polar bodies is suitable due to the great proportion of aneuploidies of maternal origin. In the majority of cases, the additional chromosome is of maternal origin. The majority of aneuploidies take place in the first meiotic division [3,4].
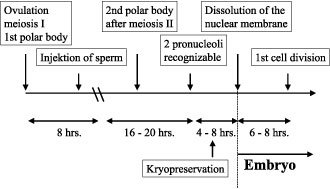
Figure 1. Time schedule of polar body formation and fusion of pronuclei.
|
|
|
|



 |
Number 27
VOL. 27 (2), 2024 |
Number 27
VOL. 27 (1), 2024 |
Number 26
Number 26 VOL. 26(2), 2023 All in one |
Number 26
VOL. 26(2), 2023 |
Number 26
VOL. 26, 2023 Supplement |
Number 26
VOL. 26(1), 2023 |
Number 25
VOL. 25(2), 2022 |
Number 25
VOL. 25 (1), 2022 |
Number 24
VOL. 24(2), 2021 |
Number 24
VOL. 24(1), 2021 |
Number 23
VOL. 23(2), 2020 |
Number 22
VOL. 22(2), 2019 |
Number 22
VOL. 22(1), 2019 |
Number 22
VOL. 22, 2019 Supplement |
Number 21
VOL. 21(2), 2018 |
Number 21
VOL. 21 (1), 2018 |
Number 21
VOL. 21, 2018 Supplement |
Number 20
VOL. 20 (2), 2017 |
Number 20
VOL. 20 (1), 2017 |
Number 19
VOL. 19 (2), 2016 |
Number 19
VOL. 19 (1), 2016 |
Number 18
VOL. 18 (2), 2015 |
Number 18
VOL. 18 (1), 2015 |
Number 17
VOL. 17 (2), 2014 |
Number 17
VOL. 17 (1), 2014 |
Number 16
VOL. 16 (2), 2013 |
Number 16
VOL. 16 (1), 2013 |
Number 15
VOL. 15 (2), 2012 |
Number 15
VOL. 15, 2012 Supplement |
Number 15
Vol. 15 (1), 2012 |
Number 14
14 - Vol. 14 (2), 2011 |
Number 14
The 9th Balkan Congress of Medical Genetics |
Number 14
14 - Vol. 14 (1), 2011 |
Number 13
Vol. 13 (2), 2010 |
Number 13
Vol.13 (1), 2010 |
Number 12
Vol.12 (2), 2009 |
Number 12
Vol.12 (1), 2009 |
Number 11
Vol.11 (2),2008 |
Number 11
Vol.11 (1),2008 |
Number 10
Vol.10 (2), 2007 |
Number 10
10 (1),2007 |
Number 9
1&2, 2006 |
Number 9
3&4, 2006 |
Number 8
1&2, 2005 |
Number 8
3&4, 2004 |
Number 7
1&2, 2004 |
Number 6
3&4, 2003 |
Number 6
1&2, 2003 |
Number 5
3&4, 2002 |
Number 5
1&2, 2002 |
Number 4
Vol.3 (4), 2000 |
Number 4
Vol.2 (4), 1999 |
Number 4
Vol.1 (4), 1998 |
Number 4
3&4, 2001 |
Number 4
1&2, 2001 |
Number 3
Vol.3 (3), 2000 |
Number 3
Vol.2 (3), 1999 |
Number 3
Vol.1 (3), 1998 |
Number 2
Vol.3(2), 2000 |
Number 2
Vol.1 (2), 1998 |
Number 2
Vol.2 (2), 1999 |
Number 1
Vol.3 (1), 2000 |
Number 1
Vol.2 (1), 1999 |
Number 1
Vol.1 (1), 1998 |
|
|

